Search equation
Please enter the reactant or product to start the search
Cl2 + C6H5CH3 → HCl + C6H5CH2Cl | , Phản ứng thế
Cl2 | chlorine | solid + C6H5CH3 | | solid = HCl | hydrogen chloride | solid + C6H5CH2Cl | Benzyl chloride; (Chloromethyl)benzene; NCIC-06360; RCRA waste number P-028; omega-Chlorotoluene; Chloromethylbenzene; alpha-Chlorotoluene; Phenyl(chloro)methane; 1-(Chloromethyl)benzene; 1-Chloromethylbenzene; Chloro(phenyl)methane; Chlorophenylmethane | solid | Temperature: temperature, Other Condition excess chlorine
Introduce
Detailed information about the equation
Reaction conditions when applied Cl2 + C6H5CH3
- Catalyst: not available
- Temperature: normal
- Pressure: normal
- Other conditions: light
Reaction process Cl2 + C6H5CH3
Process: Cl2 reacts with C6H5CH3
Note: not available
The result of the reaction Cl2 + C6H5CH3
The phenomenon: updating...
Detailed information on the reactants
Information about Cl2 (chlorine)
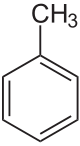
Information about C6H5CH3
Detailed information about the products of the reaction
Information about HCl (hydrogen chloride)
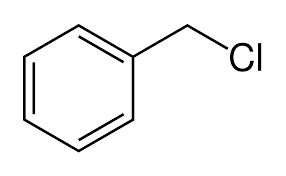
Information about C6H5CH2Cl (Benzyl chloride; (Chloromethyl)benzene; NCIC-06360; RCRA waste number P-028; omega-Chlorotoluene; Chloromethylbenzene; alpha-Chlorotoluene; Phenyl(chloro)methane; 1-(Chloromethyl)benzene; 1-Chloromethylbenzene; Chloro(phenyl)methane; Chlorophenylmethane)
Total rating:
Rating: / 5 star
The equations for preparation Cl2
Catalyst
normal
Temperature
room temperature
Pressure
normal
Other conditions
normal
Catalyst
normal
Temperature
500
Pressure
normal
Other conditions
normal
Catalyst
normal
Temperature
100
Pressure
normal
Other conditions
normal
The equations for preparation C6H5CH3
Catalyst
normal
Temperature
normal
Pressure
normal
Other conditions
Pd
+ → +
Catalyst
normal
Temperature
heated
Pressure
normal
Other conditions
normal
Catalyst
normal
Temperature
normal
Pressure
normal
Other conditions
normal
Interesting facts about chemistry you may not know
Interesting facts about hydrogen - the lightest element in the periodic table.
Hydrogen is the first element in the periodic system table. Hydrogen is known to be the lightest of all, the most abundant in the Universe, the essential element for life
View moreInteresting facts about helium
Helium is the first rare gas element in the periodic system table. In the Universe, it ranks second in abundance after elemental hydrogen.
View moreInteresting facts about lithium
Lithium is the alkali metal element, located in the third cell in the periodic table system. Lithium is the lightest of all solid metals and can cut a knife.
View moreInteresting Facts About Beryllium
Beryllium is the lightest alkaline earth metal. Beryllium is found in precious stones such as emeralds and aquamarine. Beryllium and its compounds are both carcinogenic.
View moreInteresting Facts About Carbon
Carbon is the non-metallic element in the sixth cell in the periodic system table. Carbon is one of the most important elements in all life, it is also known as the back.
View more